Objective:
1. To carry out some simple chemicals test in order to distinguish between aldehydes, ketones and phenols
2. To study the properties of aldehydes, ketones and phenols.
3. To identify the unknowns A, B, C, D and E.
Introduction:
Part I : Reaction of Aldehydes and Ketones
The carbonyl group is C=O and any compound containing this group that can be described as a carbonyl compound. Carbonyl compounds fall into two main classes: aldehydes and ketones on the one hand and carboxylic acids and their derivatives on the other hand. The characteristic reactions of the aldehydes and ketones are addition and oxidation reactions occurring at the unsaturated carbonyl group. With the same reagent, aldehydes usually react faster than ketones, mainly because there is lees crowding at the carbonyl carbon and the steric effect. Aldehydes are also more easily oxidized than ketones. The carbonyl and other compounds investigated in this experiment are tested in each of the following ways:
A) Chromic Acid (H2CrO4)
Chromic acid is a strong oxidant. Aldehydes are oxidized to carboxylic acids by chromic acid. The Cr6+ in the chromic acid which is orange, then is reduced to Cr3+ which is green/blue. Ketones are not oxidized by chromic acid.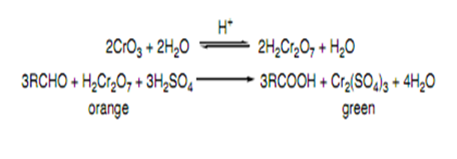 B) Tollen’s Test (Ag(NH3)2+ / OH-
B) Tollen’s Test (Ag(NH3)2+ / OH-
Tollen’s reagent (Ag(NH3)2+ / OH- is a weak oxidant. Aldehydes are readily oxidized to carboxylic acids by Tollen’s reagent to produce a silver mirror on the inside of a clean test tube. Ketones are not oxidized by Tollen’s reagent.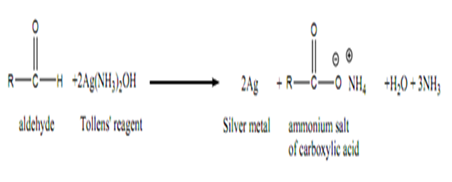
C) Fehling’s solution
Fehling’s solution is an oxidizing agent. It is prepared by mixing equal part of Fehling’s solution I (copper(II) sulfate) and Fehling’s solution II (sodium potassium tartate and sodium hydroxide). Aldehydes are easily oxidized to carboxylic acid by Fehling’s solution and will reduce the cupric ion which complexed with tartate ion to cuprous oxide. A positive result is indicated by the formation of a brick red precipitate. Ketones are not oxidized by Fehling’s solution.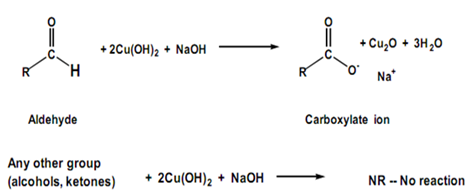
D) 2,4-dinitrophenylhydrazine (DNP Test or Brady’s Reagent)
2,4-dinitrophenylhydrazine (Brady’s reagent) is an important reagent related to hydrazine. Most aldehydes and ketones very readily with this reagent to give the yellow orange and red precipitates of 2,4-dinitrophenylhydrazones. Unconjugated aldehydes and ketones give precipitates toward the yellow while conjugated compound tend to be deeper colour of red. The conversion of aldehydes and ketones into hydrazone is an example of the addition-elimination reaction occurring at the unsaturated carbonyl group.
E) Iodoform Test
Iodoform test can be used for the detection of acetalaldehyde and all methyl ketone which have the formula: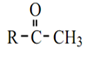
Iodoform, CHI3 is a yellow solid with a strong medicinal smell. Iodoform will precipitate out of a mixture of methyl ketone, iodine and base.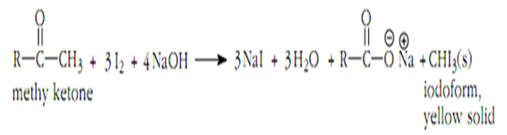 For acetaldehyde, the following reaction shows the formation of iodoform:
For acetaldehyde, the following reaction shows the formation of iodoform: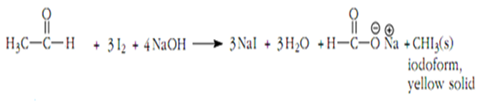
Compounds that are easily oxidized to acetaldehyde and methyl ketones also give a positive iodoform test. Only ethanol can be oxidized to acetaldehyde and secondary alcohol that have the general formula CH3CH(OH)R can be oxidized to methyl ketones.
Part II: Reactions of Phenols
Compounds in which a hydroxyl group is bonded to an aromatic ring are called phenols. Alcohols and phenols are similar in some ways, but there are enough differences so that they are considered different functional groups. One major difference is that phenols are typically about a million time more acidic than alcohols. We shall focus on chemical reactions that can help to distinguish phenols from alcohols.
A) Solubility of Phenols
The presence of a hydroxyl group in phenols permits hydrogen bonding between them and the similar substance water H-OH. This leads to appreciable water solubility of phenols. If non polar groups like alkyl groups are attached to the aromatic ring, the water solubility of the phenols decreases.
B) Acidity of Phenols
Most phenols are weaker acids than carboxylic acids and stronger than alcohols. When phenols react with a base, the phenol is converted phenoxide anion. The phenoxide anion is more soluble in water than the corresponding phenol. Consequently, if a water-insoluble phenol is treated with an aqueous solution of a base that is strong enough to convert most of the phenol to phenoloxide anion, that phenol will dissolve in the aqueous base as the phenoxide salt. None of the above-mentioned bases is strong enough to convert a substantial amount of a typical alcohol into an alkoxide anion which would cause a water-soluble alcohol to dissolve as its alkoxide anion.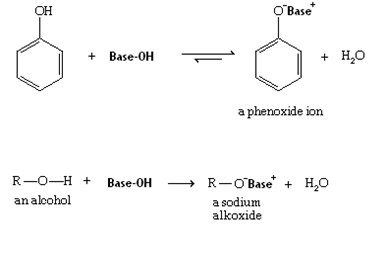
C) Reaction with Bromine Water
The hydroxyl groups of phenols activate the ring to electrophilic substitution, so that reaction occurs under very mild conditions. With bromine water and phenol, the product is 2,4,6-tribromophenol, which has such a low solubility in water and appears as a white precipitate.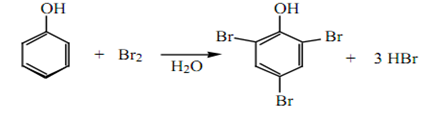
D) Ferric Chloride Test
Phenols give a colouration (pink, green or violet depending on the structure of the phenol) with ferric chloride. This is due to the formation of certain coordination complexes with the iron. Ordinary alcohols do not react. This test may be used to distinguish most phenols from alcohols.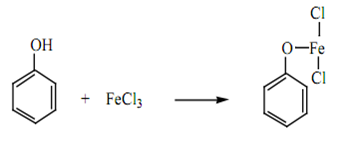
Apparatus: test tubes
Materials: chromic acid, Tollen’s reagent, Fehling’s solution I, Fehling’s solution II, 2,4-dinitrophenylhydrazine, iodine solution, 1,4-dioxane, acetophenone, 3-pentanone, acetaldehyde, benzaldehyde, isopropyl alcohol, 1-propanol, unknown A, unknown B, unknown C, 5% sodium hydroxide, 5% sodium carbonate, 5% sodium bicarbonate, bromine water, 1% ferric chloride, phenol, 2-naphthol, unknown D, unknown E
Procedure:
Part I: Reactions of aldehydes and ketones
A) Chromic acid test
1. To 1ml of acetaldehyde, benzaldehyde, acetophenone, 3-pentanone, isopropyl alcohol and 1-propanol in separate dry test tube, 1ml of chromic acid was added. Any changes in colour was observed.
2. The above procedurs was repeated with unknown A,B and C.
B) Tollen’s test (Ag(NH3)2+/OH-)
1. Preparation: Tollen’s reagent is prepared freshly before it is needed. 2 drops of 3M of NaOH was added to 10ml of 0.2M AgNO3. Then 2.8% ammonium hydroxide was added dropwise with stirring until the precipitate of AgNO3 just dissolves. Allow time for the solid to dissolve.
2. Cleaning test tube: A set of test tubes were cleaned by adding 5-10ml of 3M NaOH to each and heating them in water bath while preparing the Tollen’s reagent.
3. Carrying out the test: The NaOH was emptied and rinse them with distilled water and 2ml of Tollen’s reagent was added to 1ml of acetaldehyde, benzaldehyde, acetophenone, 3-pentanone, isopropyl alcohol and 1-propanol in separate test tube. Set the tubes aside with agitating the contents for few minutes. Warm the mixture briefly on water bath if no reaction.
4. Step 3 was repeated with unknown A, B and C and observations were made.
5. Destroying the reagent: Add a few drops of HNO3 to Tollen’s reagent after done the test.
C) Fehling’s test
1. Preparation: Equal volume of Fehling’s solution I (copper(II) sulfate) and Fehling’s solution II (sodium potassium tartrate and sodium hydroxide) were mixed.
2. To 1ml of acetaldehyde, benzaldehyde, acetophenone, 3-pentanone, isopropyl alcohol and 1-propanol in separate dry test tube, 6 drops of Fehling’s solution were added. The test tubes were heated in a hot water bath for several minutes. Reddish precipitate of Cu2O will indicate a positive test for aldehyde.
3. Step 2 was repeated with the unknown A, B and C and observations were made.
D) 2,4-dinitrophenylhydrazine
1. Preparation: 4g of 2,4-dinitrophenylhydrazine was dissolved in 8ml of concentrated H2SO4. The mixture was cooled and added with 90ml of methanol and 10 ml of water.
2. To 1ml of acetaldehyde, benzaldehyde, acetophenone, 3-pentanone, isopropyl alcohol and 1-propanol in separate dry test tube, 2ml of Brady’s reagent was added and were shaked vigorously. Warm and allow to stand for 5-10 minutes if no precipitation form immediately. A crystalline precipitate indicates the presence of a carbonyl compound.
3. Step 2 was repeated with the unknown A, B and C and observations were made.
E) Iodoform test
1. Preparation: 20g of KI and 10g of iodine crystals were dissolved in 100ml of water.
2. To 1ml of acetaldehyde, benzaldehyde, acetophenone, 3-pentanone, isopropyl alcohol and 1-propanol in separate dry test tube, 2ml of water was added. For insoluble compound, 1,4-dioxane was added one drop at a time with swirling until a homogenous solution is formed. Then, 2ml of 5% NaOH solution was added with swirling and potassium iodide-iodine reagent was added dropwise until a deep red-brown colour persists. The mixture were heated in water bath of 60 °C at least two minutes. The test tubes were removed from water bath and sufficient 5% NaOH was added dropwise to cause the disappearance of red brown colour. Cool down to room temperature and record whether a yellow precipitate forms within 15 minutes.
3. Step 2 was repeated with the unknown A, B and C and observations were made.
Part II: Reactions of phenols
A) Solubility of phenols
1. To 0.05g of phenol and 2-naphthol in separate dry test tube, 2ml of water was added. Mix and observe whether the compound is completely soluble, partially soluble and insoluble.
2. The above procedure was repeated with unknown D and E. Observation were made.
B) Acidity of phenols
1. To 0.05g of phenol and 2-naphthol in separate dry test tube, 2ml of NaOH, Na2CO3, NaHCO3 were added. Mix and observe whether the compound is completely soluble, partially soluble and insoluble.
2. The above procedure was repeated with unknown D and E. Observation were made.
C) Reaction with bromine water
1. 0.05g of phenol and 2-napthol were dissolved in 2ml of dilute HCl in separate dry test tube and bromine water was added with shaking until the yellow colour persists. Observation was made.
2. The above procedure was repeated with unknown D and E. Observation were made.
D) Ferric chloride test
1. 0.05g of phenol and 2-napthol were dissolved in water (or mixture of water and ethanol if the compound is not water soluble) in separate dry test tube and 1% of ferric chloride was added dropwise. Shake and observe any colour changes.
2. The above procedure was repeated with unknown D and E. Observation were made.
Results:
Part I: Reaction of Aldehydes and Ketones
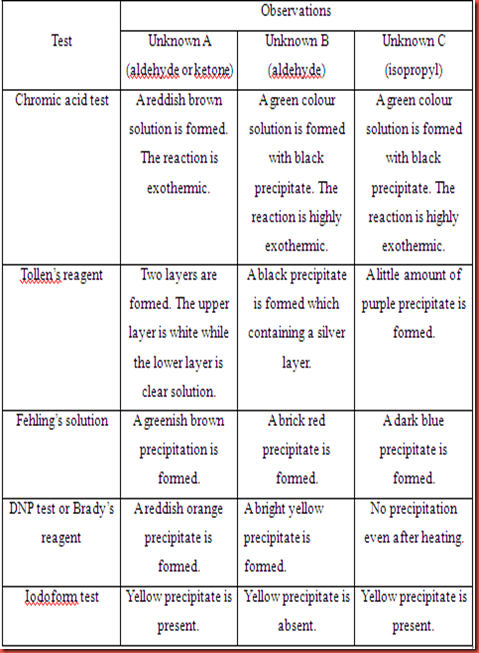
Table 1.1 Chemical tests on unknown A, B and C.
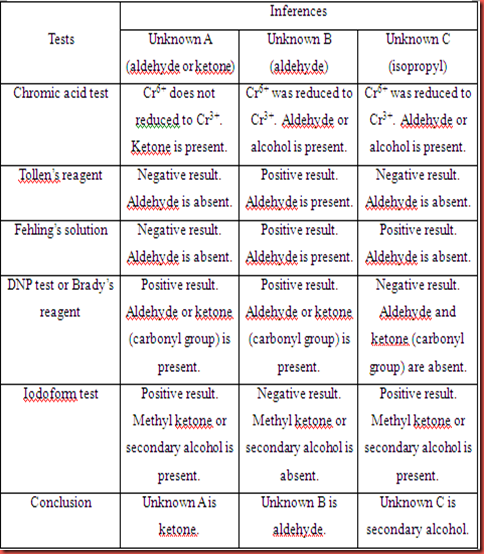
Table 1.2 Inferences based on observation for unknown A,B and C.Part II : Reactions of phenols
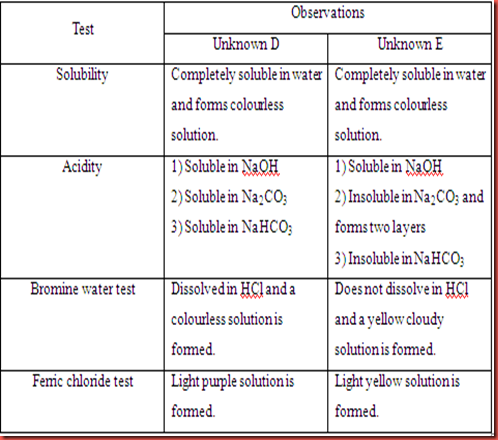
Table 2.1 Chemical tests on Unknown D and E
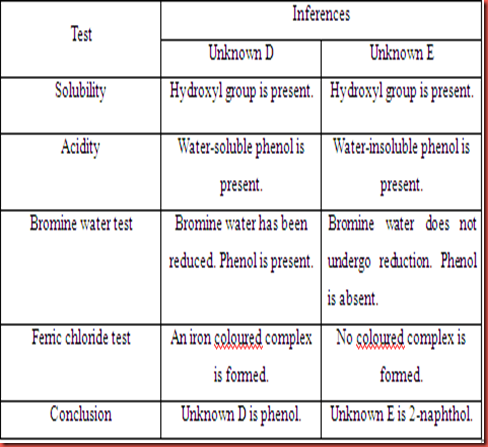
Table 2.2 Inferences based on observation for unknown D and E.
Discussion:
Chromic acid test can be used to differentiate aldehyde and ketone. Since chromic acid is a strong oxidizing agent. It can oxidize alcohols and aldehydes to form carboxylic acid but it will not oxidize ketone. The change in colour from orange solution to green solution shows a positive test which due to the change in oxidation state of the chromic metal. The chromic acid will be reduced from Cr6+ (orange) to Cr3+(green) which undergoes reduction. Tollen’s reagent is a test used to identify a carbonyl compound whether the compound is aldehyde or ketone. Aldehyde is readily oxidized to form carbocylic acid but ketone is not. The positive result in Tollen’s reagent is the formation of silver mirror or black precipitate in the solution. For Fehling’s test, the functional group of aldehyde can be tested via reduction of deep blue solution of copper(II) to a formation of brick red precipitate. In DNP test, Brady’s reagent is usually used to test on aldehydes and ketones. The formation of yellow, orange or red precipitate give the positive result to the compounds with carbonyl groups. Besides, iodoform test is functioned as the test for α-methyl group present in aldehydes, ketones and secondary alcohols. The positive result can be seen through the formation of yellow precipitate in the particular test.
For unknown A, it gave negative results in the chromic acid test and Tollen’s test. The unknown A cannot be oxidized by the chromic acid and Tollen’s reagent. In Fehling’s solution, there is a formation of greenish brown precipitate in this test. The unknown A in this test gave negative result because unknown A cannot shows positive test in the first two tests respectively. So, it is believed that the formation of the precipitate is due to the reaction between the unknown A and the metal in Fehling’s reagent. Unknown A shown a positive result in DNP test which the reddish orange precipitate was formed. This is because the C=O functional group is exists in the unknown A. In iodoform test, the present of yellow precipitate indicated that the positive test for iodoform test. This is shows that unknown A consists of α-methyl group in its structure. The unknown A is predicted has ketone group, with more specified is predicted as acetophenone.
In chromic acid test, unknown B gave a positive result which orange solution is turned to green solution. This is shows that unknown B is oxidized by the chromic acid. So, the following test Tollen’s test and Fehling’s test also shows the positive results. The formation of silver mirror and brick red precipitate are formed in the Tollen’s test and Fehling’s test respectively. In DNP test, unknown B also gave the positive result which the bright yellow precipitate is formed. This is indicates that the unknown B has the functional group of C=O in its compound. However, the unknown B did not give a positive result in the iodoform test. So, we can predict that the α-methyl group is absent in the compound. Unknown B is predicted as benzaldehyde which is categorized into aldehyde group.
The positive result in chromic acid test for unknown C shows that the reduction of Cr6+ to Cr3+ take places in the reaction. Unknown C is considered as undergoes oxidation via chromic acid test since chromic acid is a strong oxidizing agent. The unknown C cannot be oxidized by Tollen’s reagent and Fehling’s solution. Unknown C gave negative result in both tests which no formation of silver mirror in Tollen’s test and no brick red precipitate in Fehling’s solution. It is also show a negative result in DNP test due to no precipitate form. This is shows that unknown C does not consist of α-methyl group in its compound. Unknown C is predicted as isopropyl alchol which is under the category of alcohol.
There are four tests to differentiate phenolic compound which include solubility, acidity, bromine water test, and ferric chloride test. Solubility is used to differentiate the compound is completely soluble, partially soluble or non soluble in water. Solubility of compounds is the tendency of the compound to form the hydrogen bonding with the water molecules. The formation of homogeneous solution after the compound dissolved completely in water indicates the positive result. In the acidity of phenolic compound test, the strong base will tend to react with the compound by deprotonating the phenolic compound. Normally, sodium hydroxide and sodium carbonate are strong enough bases to dissolve most water insoluble phenols instead of sodium bicarbonate. The degree of dissociation can be shown in the solubility of the phenolic compound in the strong base solution. Positive result is indicated by the tendency of phenolic compound dissolve in all the base solution. In bromine water test, the decolouration of yellow solution shows a positive test which is followed by the formation of white precipitate. In ferric chloride test, phenol tends to react with ferric chloride to give a coloured complex. The change of colour in the solution shows the positive result.
Unknown D is soluble in the water and tends to form a homogeneous solution. Similarly, it is also dissolved completely in sodium hydroxide, sodium carbonate, and sodium bicarbonate. The acidity of unknown D can be shown in the solubility in sodium hydroxide, sodium carbonate, and sodium bicarbonate. Unknown D is soluble in all base solution although it is a weak acid. However, it is a water soluble phenol. So, it can soluble in the three base solutions. In the bromine water test, the unknown D had decolourized the yellow bromine water. This is shows that unknown D is undergoes bromination to form a brominated phenolic compound. In the ferric chloride test, the solution turns to purple solution after ferric chloride was added. This is shows that unknown D is not an ordinary alcohol. Thus, unknown D is predicted as phenol.
For unknown E, it is soluble in water and form homogenous solution. However, unknown E is only soluble in sodium hydroxide but not in sodium carbonate and sodium bicarbonate either. This is shows that unknown E is a water-insoluble phenol and is categorized as a weak acid. In bromine water test and ferric chloride test, the colours of bromine water and ferric chloride were remained unchanged in each test. So, this is means that unknown E does not react with bromine and ferric chloride. As a result, the unknown E is considered as ordinary alcohol.
Precaution steps:
1. Phenolic compounds are corrosive and cause severe chemical burns on contact.
2. When phenol is applied directly to the skin, a white covering of precipitated protein forms. This soon turns red and eventually sloughs, leaving the surface stained slightly brown. If phenol is left on the skin, it will precipitate rapidly and lead to cell death and gangrene. Phenol appears to have local anesthetic properties and can cause extensive damage before pain is felt.
3. When skin comes in contact with phenol, irrigate exposed area with large volume of water.